
|
|

DC Diana Hampe
Investigations on the reactivity of metal-alkylidene-dihydropyridines
towards heterocumulenes (and other electrophiles)
-
Deprotonation of 2- or 4-alkylpyridines in Cα-position leads to the corresponding dihydropyridine
anions which have at least two reaction centers ? the Cα-atom or the ring nitrogen, respectively. There is one
more reactive atom if an azomethine or a hydrazone substituent is bound in Cα-position ? the former azomethine
carbon or the former NH nitrogen, respectively.
These ambident anions are the starting point for several interactions and reactions with a variety of electrophiles,
but the emphasis lays on different heterocumulenes.
The main points can be summarized as follows:
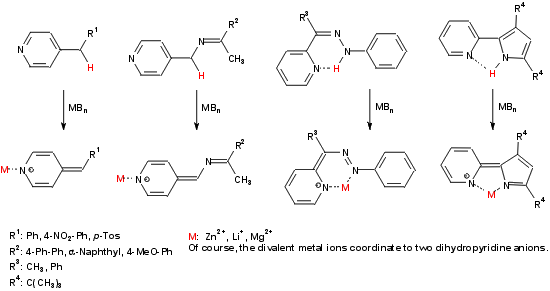
Reversible and irreversible fixation of heterocumulenes (CO2, CS2, PhNCO, PhNCS, ketenes, allenes, ?)
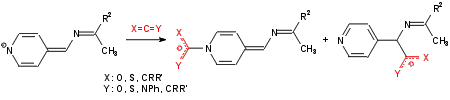
Activation of heterocumulenes in transfer reactions to C-H-acid compounds (1,3-diketones, β-ketoesters,
cyanacetic acidesters, malononitrile, ?).
The reaction of 4-alkyliden-1,4-dihydropyridines substituted by an azomethine moiety with carbondioxide is modeled
after a biological example: pyridoxamine, a pyridine derivative of the vitamine B6-complex which facilitate the
decarbxylation of aminoacids via a dihydropyridine.
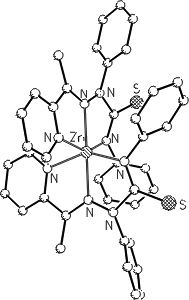
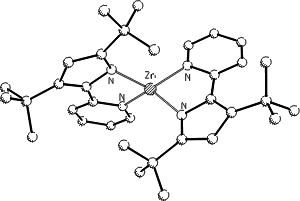
The selection of the metal ions (Zn, Mg) is also inspired by nature: zinc in the enzyme carboanhydrase and magnesium
in biotin-dependent enzymes (involved in carbon dioxide fixation and transfer).
Further interesting reactions are:
Synthesis of new heterocycles (thiazoline-thiolates, imidazolidine-thiones, triazolidine-thiones)
Synthesis of new β-and γ-aminoalcohols (chiral) by reaction with aldehydes/ketones or epoxides, respectively.
List of Publications
1,3-Thiazoline-5-thiolates and 1,3-Thiazole-5(2H)-thiones by [3+2]-Cycloaddition of Carbon Disulfide to Metal 4-Alkylidene-4H-pyridin-1-ides.
D. Hampe, H. Görls and E. Anders*, Eur. J. Org. Chem. 2005, 21, 4589-4600.
Metal 4-Alkylidene-4H-pyridin-1-ides and 2H-Imidazol-4-ones from Novel Highly Substituted Azomethines. D. Hampe, W. Günther, H. Görls and E. Anders*, Eur. J. Org. Chem. 2004, 4357-4372.
|
|







